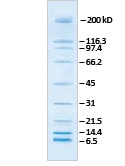
The blue-stained WesternC standard bands fluoresce in the red range (10, 15, 20, 37, 100, 150, 250 kD) and the pink bands (25, 50, 75 kD) fluoresce in the green range the fluorescent blot images were taken using Bio-Rad’s VersaDoc™ MP 5000 imaging system and overlaid. The WesternC protein standards are shown below (left) on a blot developed via chemiluminescence (using the Precision Protein StrepTactin-HRP conjugate) and are shown (right) on a blot developed via fluorescence. Visualization of all ten bands of the standard on your membrane indicates that proteins have transferred, and the blot can be moved to the next step in the western blotting workflow.Īfter developing the blot via chemiluminescence using Precision Protein ® StrepTactin-HRP or -AP conjugates or fluorescence (no conjugate needed), one can then confirm the target protein(s) by molecular weight. The WesternC standards are shown below on a PVDF membrane after a transfer using the Trans-Blot ® Turbo™ transfer system, which enables protein transfers in as little as three minutes. Additionally, the dual-color feature of the standard provides a quick way to look at the orientation of your blot. WesternC protein standards also enable easy monitoring of transfer process, providing a broad molecular weight range, from 10 to 250 kD. Once the ten standard bands are separated and visualized, you can move onto the next step of the workflow. The WesternC standards are shown below after electrophoretic separation on a 4–20% Mini-PROTEAN ® TGX™ gel, which allows protein separation in as little as 15 minutes. WesternC protein standards have dual-colored prestained bands that enable you to monitor electrophoretic separation. Monitor Your Western Blotting Workflow with Precision Plus Protein WesternC standards The WesternC standards have dyes that fluoresce under green and red wavelengths. *StrepTactin-HRP or StrepTactin-AP conjugate is needed for colorimetric and chemiluminescent blot development, but not for fluorescence development. The following workflow illustrates the use of Precision Plus Protein WesternC standard with a suite of Bio-Rad products, all of which minimize the time to results for your western blotting workflow. This protein standard can be used regardless of your detection method* (colorimetric, chemiluminescent or fluorescent). This protein standard is unique in that it offers the ability to visualize and monitor electrophoretic separation, assess transfer of proteins to your membrane, and visualize and estimate the molecular weight of your target after blot development with a StrepTactin-HRP or AP conjugate. Bio-Rad’s Precision Plus Protein WesternC standard enables you to check the progression and success of all the major steps in the western blotting process. Thus, having a tool to monitor each of the major steps in the western blotting workflow reduces the chances of finding out at the very end of the workflow that an experiment was not successful. The whole western blotting workflow can take up to three days to complete. So I don't understand how the protein could either come out the other side, or not go in at all, but the ladder be fine.Western blotting, although a commonly used technique, involves a time-consuming multi step process. 
Would one of the two observations listed above suggest something that would allow the pre-stained ladder to transfer successfully, but not the protein? The ladder transfered evenly from 250 all the way down to 15 kD. So there is no protein, but the curious thing is the pre-stained ladder came out fine. pvdf thread in this forum that not soaking in water long enough can prevent protein transfer in PVDF)Ģ) His transfer chamber, when put at the same voltage I normally run (30v) went up to something like 260 mA which is about 4 times higher than the normal mA I see with my mini-transfer chamber. I told him to pre-wet in methanol, which we did, but instead of soaking in water for 5 minutes as per the instructions, he just rinsed and put in the transfer sandwich. When he was putting together the transfer, he dunked the pvdf directly in water and acted surprised when it didnt hydrate. Two suspicious things:ġ) This professor always used nitrocellulose and we use PVDF (hybond-P). In fact, except for the sample preparation in sample buffer, he pretty much did everything. I ran the big gel with professor who does them a lot. However, I wanted to get more resolution so for this particular western I had run a "big" gel. In fact, I had run these exact samples already twice and it worked fine. I run mini-gels all the time and never have problems. I encountered something strange the other day with my western blot:Īfter transfer, the pre-stained ladder (bio-rad) showed up fine, but there was no protein whatsoever on the blot stained with Ponceau.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
